Background
One ongoing point of contention among brewers is what benefits, if any, result from rehydrating dry yeasts according to manufacturers’ recommendations, as opposed to simply adding the yeast directly to the fermenter. On the technical level, there would appear to be a consensus for a substantial reduction in viability when rehydrating in wort, with most sources claiming a reduction on the order of 50%. [1, 2, 3, 4] There exists a substantial body of anecdotal evidence, however, maintaining that rehydration offers no perceptible benefits. I already have some data on the effect of pitching rate on beer flavor, and I think it’s reasonable to infer that a similar reduction in pitching rate would result in similar effects, regardless of whether that yeast was originally a dry or liquid culture. To that end, I decided to do a series of basic viability estimates using dry yeast rehydrated under various conditions.
Experimental Setup
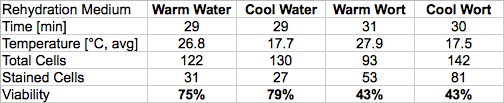
Four cell counts were conducted, using Safale US-05 yeast obtained from a 500 g sachet that had been stored cold and was opened the same day. The yeast, according to the stamp on the packaging, was 176 days old at that point. Assuming that it was initially nearly 100% viable cells, and lost 4% per month, its viability should therefore have been roughly 75-80%. One level one-third teaspoon measure (approx. 1.3 g) was rehydrated in 100 mL of each of four different media: tap water and wort at both typical ale pitching temperature (~18°C) and rehydration temperature (~33°C). The water used is fairly low in mineral content; it was tested the following day at 50 ppm CaCO3 total alkalinity and 100 ppm CaCO3 total hardness. The wort used had a gravity of 11.5°P.
After resting loosely covered for half an hour, each sample was capped and shaken to homogenize it. 1.00 mL was then pipetted into 49.0 mL of a 0.02% methylene blue solution and agitated for one minute before a drop was placed on a Neubauer cytometer plate for counting. With the exception of the methylene blue concentration, I use the White Labs protocol for counting – I’ve found a 0.1% solution to be far too dark for effective discrimination of viable and non-viable cells.
Results
With an average total count of 122 cells, the cellular density for the dry yeast works out to 22.9 billion/gram, which correlates well with the established figure of 20 billion/gram considering that the yeast was dispensed by volume rather than mass. The viability estimates for the samples rehydrated in water are also remarkably close to the predicted values, although interestingly enough, the rehydration temperature did not appear to have a substantial effect on viability in either medium. Setting aside temperature as a variable, then, the average viability after rehydrating in wort is 56% of that for the samples rehydrated in water.
Based on these results, I’m confident that the conventional wisdom regarding dry yeast is essentially correct, and that a substantial reduction in the effective pitching rate would result from rehydrating in wort. Additional trials would need to be conducted to determine conclusively whether or not the rehydration temperature is as critical as the manufacturers’ recommendations suggest.
Sean,
Nice experiment. I meant to do the same experiment ever since I got my microscope and even bought dry yeast. That was more than a year ago.
One thing to keep in mind is that the methylene blue staining can be misleading. I had old yeast samples where almost none of the cells were stained but when I streaked it on a plate nothing grew.
Kai
Thank you for this. It’s good stuff. Though, I probably will continue to drop the dry yeast into the wort, for a couple reasons.
First, rehydrating yeast in my circumstance, it’s just another venue to introduce contamination. Second, the head start in numbers could be temporary. For instance, in experiments involving fertilizers and tree growth, the head start is overcome in a few seasons. My presumption is that if we were to superimpose the population bell curves , they would both peak at nearly the same number.
Do you think rehydrated versus dry-pitched yeast would produce noticeably different beers?
Norm,
As with anything else in brewing, if there are sanitation concerns then those definitely need to be addressed before adding any new process. In that case, though, I would advise pitching two packs per 5 gal of average-gravity ale. I think you’re correct that the final cell counts will be similar, but off-flavors are produced by yeast stress factors during reproduction, and as I saw in the pitching rate experiment, the effect can be significant. Directly pitching a single pack of dry yeast would result in roughly the same pitching rate as the under-pitched samples in that survey.
Sean
Thanks. You polymaths are good. You are so right. I hadn’t thought of that (but now that you say it, it’s an aha moment – id est, a blinding flash of the obvious :-] ) Two yeast packs in my 5-gallon batch will work well and is darn cheap insurance to minimize off flavors.
I have a lot to learn and your posts help immensely.
Nice experiment Sean!
What would happen to the viability of the yeast cells when you first hydrate them in water and subsequently add them to the wort?
Thanks William. The main problem with rehydrating in wort is that for the first few minutes the cells can’t discriminate what crosses the cell wall, so all of the wort compounds end up in the cell. Once the cell wall has been reconstituted, the yeast can operate normally, and so there should be little to no loss of cells when pitching them into a normal-gravity wort. In high-gravity brewing, there would likely still be some cells lost to osmotic shock, though, regardless of how they had been treated prior to that point.
Sean
Why would many of the dry yeast manufacturers advice to disperse the dry yeast in water before bringing it in the wort. Doing the combination of first hydration in warm water and subsequently dilute in wort would make the results stronger.
I guess that the step in water helps to disperse the yeast cells because of the difference in osmotic pressure which gives a net in-flow of water into the yeast agglomerates. The high temperature might have to do with the stearate melting which is often used as an agglomeration agent for yeast.
The researchers at Nexel(their web site http://www.nexel.com) have used a flourescence
based cell imaging device,the “Celometer” to assess the viability of dry east strains after rehydration following the manufacturers directions. Samples from six sources gave viabilities ranging from 5 to 90%. I have no relationship to Nexel, but they have saved me alot of work by releasing their data. Nice work Sean.
[…] rehydration in tap water, do not do a starter. Sean Terrill did some rehydration experiments. http://seanterrill.com/2011/04/01/dry-yeast-viability/ __________________ What's […]
[…] for us already. Until I find the time to do the experiments myself, I'll take their word for it. example GA_googleFillSlotWithSize("ca-pub-3927874040083090", "HomeBrewTalk_Forum_336x280_BTF", 336, 280); […]
[…] Terrill, Sean. “Dry Yeast Viability.” SeanTerrill.com. Sean Terrill, 4 Apr. 2011. Web. 08 Jan. 2012. <http://seanterrill.com/2011/04/01/dry-yeast-viability/>. […]
[…] That's why I switched to Nottingham. One 11g packet gives you up to 200 billion cells (assuming you rehydrate correctly), and at 1.077 even the calculators say you need 277 billion – close enough that I don't really […]
[…] and Chris White are often mentioned and their experience and expertise sited. An interesting study HERE addresses this issue and though I find reports utilizing methylene blue vital staining dubious, […]
Could yeast nutrient be added to the cooled sterile water at any point before or after the yeast is added to increase viability?
Nutrient could definitely be added, and there’s one (GoFerm) marketed as being designed specifically for use during rehydration. It won’t impact viability though – you can’t bring a cell back from the dead.
Thanks for the reply, great work here. I wish I had stumbled upon this site earlier.
Thanks for sharing your results. In response to an earlier post you recommended pitching two sachets of dry yeast for five gallons of wort. If there are 20 billion cells per gram this would be 230 billion cells for one sachet. Wouldn’t that be enough for five gallons of wort? Is your recommendation due to the 75% viability rate? I’ve assumed one sachet was enough but with the fruity esters and lack of a large krausen I’ve wondered if one sachet if dry yeast was enough. I recently switched to making 2L starters from liquid yeast so I never tested pitching two sachets of dry yeast for five gallons of wort.
The brewer who made that comment wasn’t able/willing to rehydrate, due to sanitation concerns. In that case, yes, I would recommend double-pitching in order to compensate for the reduction in viability.
If you’re rehydrating, one sachet would be sufficient.
[…] ska jag pitcha jästen då? Här har dom utfört ett experiment på just detta och kommit fram till att man får bästa resultat om […]
[…] out the density is closer to 20B/g. See http://seanterrill.com/2011/04/01/dry-yeast-viability/ […]
[…] http://seanterrill.com/2011/04/01/dry-yeast-viability/ has some interesting experimental results, and his findings of 8 – 18 million cells per gram support MrMalty.com’s assertion that there are approximately 20 billion cells per gram of dry yeast, so in a 11.5 gram sachet, there should be approximately 230 billion cells, however Lallemand claims only 6 billion cells per gram, resulting in 69 billion cells per sachet, and in fact there are tables floating around the internet of WIDELY varying cell density. That is a HUGE discrepancy, and I use the manufacturer’s numbers personally – and the information is there if you look for it. In a White Labs vial or a Wyeast Smack Pack, there are approximately 100 billion cells. Good general working numbers. […]
[…] http://seanterrill.com/2011/04/01/dry-yeast-viability/ […]
[…] the sides of the carboy, and also the yeast will be healthier since pitching directly into the wort reduces viability by 40-50% compared to rehydrating in warm […]
[…] un famoso esperimento sul lievito secco US05 fatto da Sean Terrill (link) che dimostrebbe (il condizionale è sempre d’obbligo, parliamo di esperimenti non ufficiali) […]
[…] 69 billion cells per pack of yeast (6 billion per gram), counting exercises such as the one performed by Sean Terrill suggest a count closer to 230 billion cells per pack (20 billion per gram), which supports […]
Thanks for the great information. I noticed you are using methylene blue- I have been reading a lot of data about using trypan blue instead as it takes into account a healthy cell membrane as an important factor in uptake of the stain. I was wondering what the viability would look like using this instead of M.B.?
Kevin,
I’ve never tried trypan blue, and to be honest I no longer stain for viability after finding that all my properly-handled slurries are >90% anyway.
Cheers,
Sean
[…] http://seanterrill.com/2011/04/01/dry-yeast-viability/ […]
[…] Dry Yeast Viability […]